
BOA I.V. Constricting Band
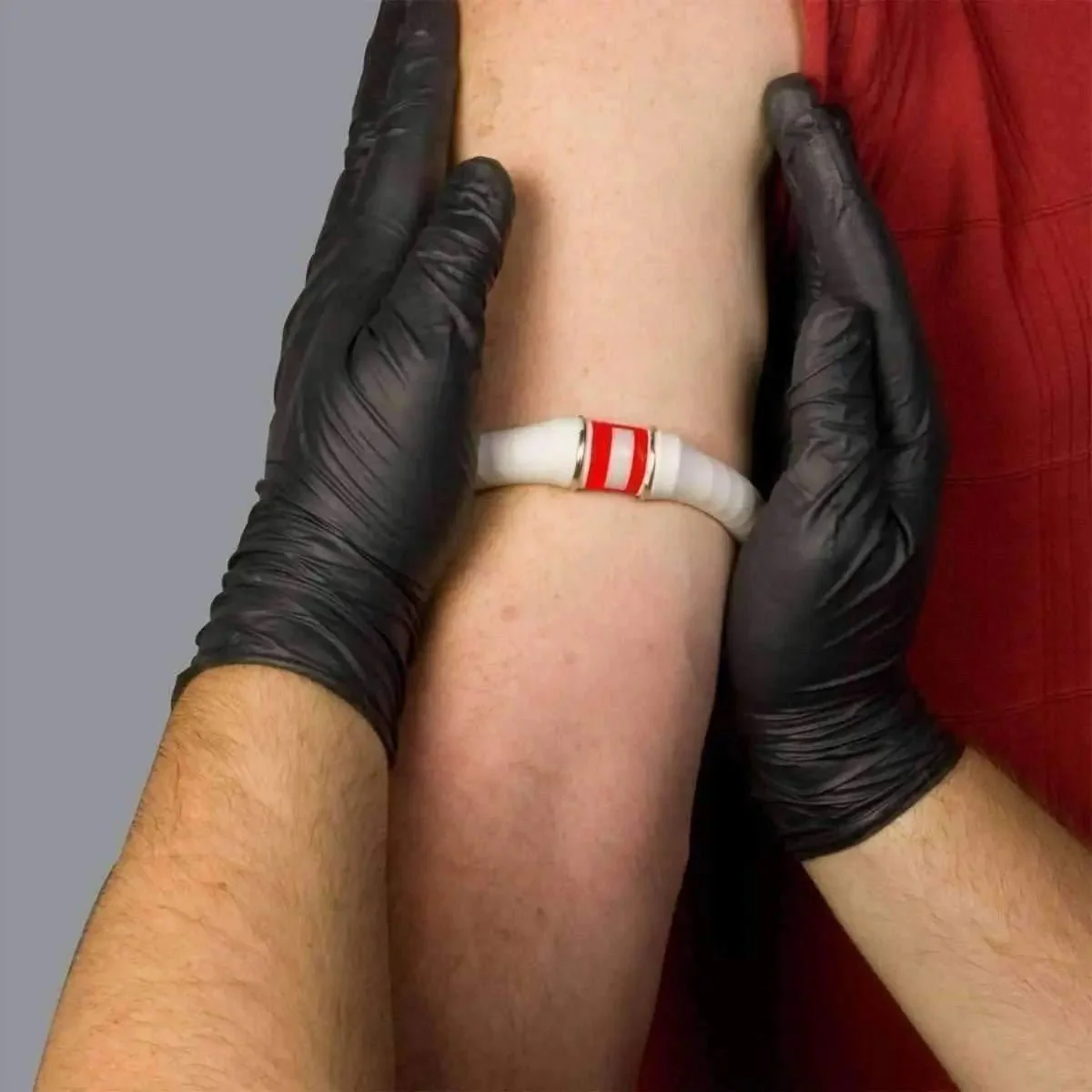
The BOA® I.V. Constricting Band by North American Rescue is a purpose-engineered, single-use venous constriction device designed for rapid peripheral IV access. Unlike flat tourniquet-style bands, the BOA uses a rolling compression mechanism that engorges distal veins while maintaining arterial flow — enabling successful cannulation in critically ill, hypovolemic, and difficult-access patients. Available in Standard (7.5"–18") and XL (10"–27") sizing.
Key Specifications
| Specification | Detail |
|---|---|
| Manufacturer | North American Rescue (NAR) |
| SKU — Standard | 30-0046 (included in Saline Lock Kit); standalone not SKU'd |
| SKU — XL | 30-0071 |
| Standard Device Length | 7.5 in. to 18 in. |
| XL Device Length | 10 in. to 27 in. (~50% longer stretch) |
| Standard Packaged Dimensions | 4 in. L × 3 in. W × 0.9 in. D |
| XL Packaged Dimensions | 4 in. H × 3 in. W × 1 in. D |
| Weight (Standard) | 1.5 oz |
| Weight (XL) | 2 oz |
| Material | Nylon / Rubber |
| Color — Standard | White |
| Color — XL | White with Red Stripe |
| Usage | Single-use, disposable |
| US Patents | 5,304,202 and 5,607,448 |
| Packaging | Rugged, vacuum-sealed |
Product Overview
Peripheral IV access is a cornerstone intervention in combat casualty care and pre-hospital emergency medicine. In hemorrhagic shock, veins collapse due to hypovolemia, making a standard rubber tourniquet-style band insufficient for reliable venous distension. The BOA® Constricting Band was originally designed for the Armed Forces specifically to address this challenge — enabling IV access on veins that traditional bands fail to visualize or cannulate.
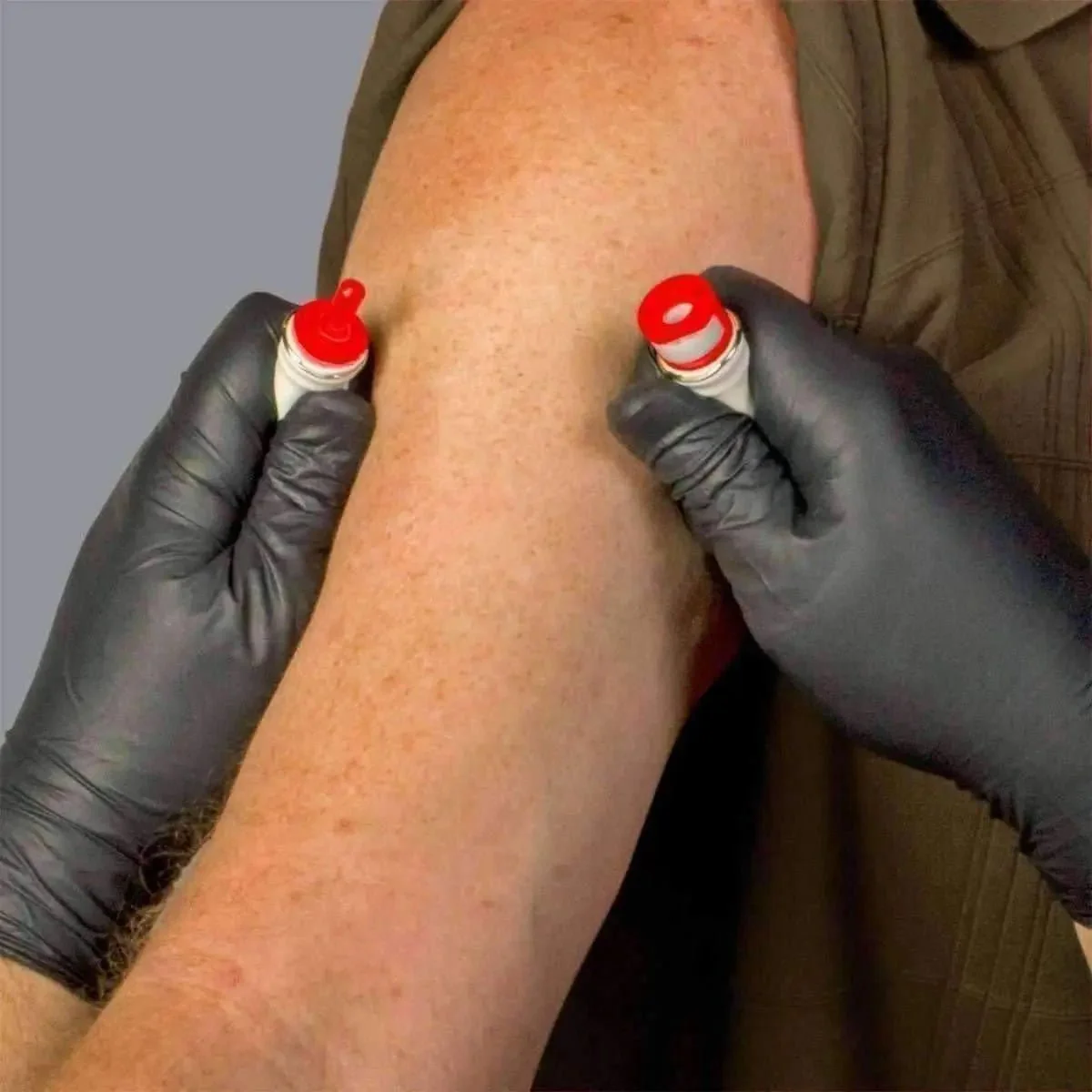
The BOA's defining feature is its rolling compression mechanism. Rather than applying uniform flat pressure (which can compress arteries as well as veins), the BOA® rolls venous blood distally into the arm, engorging peripheral veins while arterial inflow continues. This creates a visually prominent, palpable venous target even in low-volume patients. Clinical settings where this matters include: combat casualties with Class III–IV hemorrhage, diabetic patients with sclerotic or fragile veins, obese patients (BOA XL), and pediatric/elderly patients with difficult venous access.
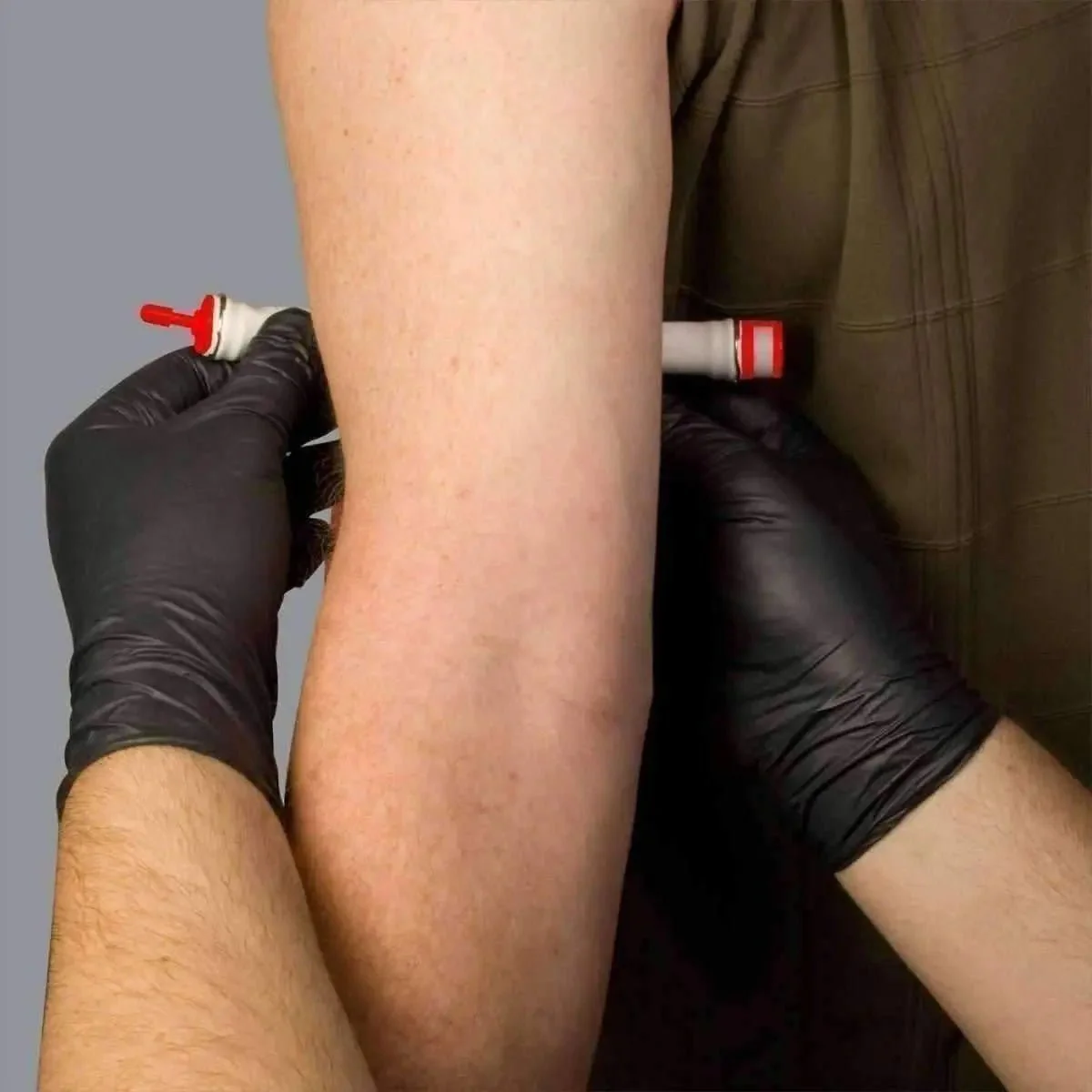
The Quick Click connector allows one-handed application and the two-finger Quick Release provides immediate removal without tool use — critical during rapid sequence interventions when both hands are needed for catheter insertion. The vacuum-sealed packaging maintains device integrity in harsh environments and enables rapid identification and access from kit pouches.
Standard vs. XL Selection Guide
| Specification | Detail |
|---|---|
| Standard BOA® | Arm circumference up to 18 in. — typical adult upper extremity |
| BOA® XL | Arm circumference up to 27 in. — larger body types, edematous extremities, upper arm access in large patients |
| Key Difference | XL stretches approximately 50% longer than Standard; white/red stripe visual differentiation |
IV Access Procedure with BOA
- Place BOA® as high on the extremity as allowable for maximum venous distension
- Apply Quick Click connector — no tying required
- Observe and palpate for venous engorgement (typically 15–30 seconds)
- Perform IV cannulation using preferred gauge catheter
- Two-finger Quick Release immediately upon successful cannulation
- Secure catheter with dressing; connect IV administration set
See also: IV/IO & Blood Transfusion Supplies
See also: IFAK Kits & First Aid
All products sourced from the actual brand manufacturer or authorized master distributors. CoTCCC recommendation status verified where applicable. Ships from MED-TAC International, Pembroke Pines, FL — clinician-founded, veteran-led, SDVOSB-certified.
Original: $149,000.00
-70%$149,000.00
$44,700.00Product Information
Product Information
Shipping & Returns
Shipping & Returns
Description
The BOA® I.V. Constricting Band by North American Rescue is a purpose-engineered, single-use venous constriction device designed for rapid peripheral IV access. Unlike flat tourniquet-style bands, the BOA uses a rolling compression mechanism that engorges distal veins while maintaining arterial flow — enabling successful cannulation in critically ill, hypovolemic, and difficult-access patients. Available in Standard (7.5"–18") and XL (10"–27") sizing.
Key Specifications
| Specification | Detail |
|---|---|
| Manufacturer | North American Rescue (NAR) |
| SKU — Standard | 30-0046 (included in Saline Lock Kit); standalone not SKU'd |
| SKU — XL | 30-0071 |
| Standard Device Length | 7.5 in. to 18 in. |
| XL Device Length | 10 in. to 27 in. (~50% longer stretch) |
| Standard Packaged Dimensions | 4 in. L × 3 in. W × 0.9 in. D |
| XL Packaged Dimensions | 4 in. H × 3 in. W × 1 in. D |
| Weight (Standard) | 1.5 oz |
| Weight (XL) | 2 oz |
| Material | Nylon / Rubber |
| Color — Standard | White |
| Color — XL | White with Red Stripe |
| Usage | Single-use, disposable |
| US Patents | 5,304,202 and 5,607,448 |
| Packaging | Rugged, vacuum-sealed |
Product Overview
Peripheral IV access is a cornerstone intervention in combat casualty care and pre-hospital emergency medicine. In hemorrhagic shock, veins collapse due to hypovolemia, making a standard rubber tourniquet-style band insufficient for reliable venous distension. The BOA® Constricting Band was originally designed for the Armed Forces specifically to address this challenge — enabling IV access on veins that traditional bands fail to visualize or cannulate.
The BOA's defining feature is its rolling compression mechanism. Rather than applying uniform flat pressure (which can compress arteries as well as veins), the BOA® rolls venous blood distally into the arm, engorging peripheral veins while arterial inflow continues. This creates a visually prominent, palpable venous target even in low-volume patients. Clinical settings where this matters include: combat casualties with Class III–IV hemorrhage, diabetic patients with sclerotic or fragile veins, obese patients (BOA XL), and pediatric/elderly patients with difficult venous access.
The Quick Click connector allows one-handed application and the two-finger Quick Release provides immediate removal without tool use — critical during rapid sequence interventions when both hands are needed for catheter insertion. The vacuum-sealed packaging maintains device integrity in harsh environments and enables rapid identification and access from kit pouches.
Standard vs. XL Selection Guide
| Specification | Detail |
|---|---|
| Standard BOA® | Arm circumference up to 18 in. — typical adult upper extremity |
| BOA® XL | Arm circumference up to 27 in. — larger body types, edematous extremities, upper arm access in large patients |
| Key Difference | XL stretches approximately 50% longer than Standard; white/red stripe visual differentiation |
IV Access Procedure with BOA
- Place BOA® as high on the extremity as allowable for maximum venous distension
- Apply Quick Click connector — no tying required
- Observe and palpate for venous engorgement (typically 15–30 seconds)
- Perform IV cannulation using preferred gauge catheter
- Two-finger Quick Release immediately upon successful cannulation
- Secure catheter with dressing; connect IV administration set
See also: IV/IO & Blood Transfusion Supplies
See also: IFAK Kits & First Aid
All products sourced from the actual brand manufacturer or authorized master distributors. CoTCCC recommendation status verified where applicable. Ships from MED-TAC International, Pembroke Pines, FL — clinician-founded, veteran-led, SDVOSB-certified.